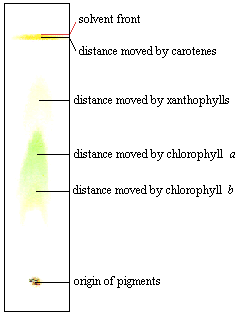
Chromatographic separation of pigments from leaves.
The chromatograpghy strip shows 4 distinct pigment bands:
Answers to questions
Calculate the Rf for each of these pigments.
Rf = (distance moved by pigment)/ (distance moved
by solvent)
= (distance form origin of pigment
to top (or middle) of pigment)/ (distance from origin of pigment to solvent
front)
-
Rf for carotenes = 9.7cm/9.8cm = 0.99
-
Rf for xanthophylls = 7.2cm/9.8cm = 0.73
-
Rf for chlorophyll a = 5.1cm/9.8cm = 0.52
-
Rf for chlorophyll b = 3.7cm/9.8cm = 0.38
What does a small Rf value indicate about the characteristics of moving
molecules?
A small Rf indicates that the moving molecules
are not very soluble in the hydrophobic (non-polar) solvent; they are larger
and/or have a greater affinity for the hydrophillic paper (they have more
polar groups) than molecules with a larger Rf.
Which are more soluble in the chromatography solvent, xanthophylls or chlorophyll
a?
Based on the Rf values, xanthophylls are more
soluble in the chromatography solvent.
Would you expect the Rf value of a pigment to change if we altered the
composition of the solvent? Why or why not?
If altering the composition of the solvent resulted
in a change the solubility of the pigment in the solvent, the Rf value
would also change.
If yellow xanthophylls were present in the extract why did the extract
appear green?
The green chlorophylls are present in larger
quantities than other pigments in both the intact leaf and the extract.
As a result, the green color of the chlorophylls masks or hides the presence
of the other pigments.
Is it possible to have an Rf value greater than 1? Why or why not?
In order to have an Rf value greater than 1 the
pigment would have to move further than the solvent. Since the pigment
is carried by the solvent an Rf greater than one is not possible.

